
Than substances with weaker intermolecular forces. In the glycerol the ball falls slowly,įigure 4.55: The higher the viscosity (red) the slower the ball moves throughĪs implied by the definition, substances with stronger intermolecular forces are more viscous Is for the ball to fall to the bottom ( Figure 4.55). Drop a small metal ball into each cylinder and note how easy it You can see this if you take a cylinder filled with water and a cylinder filled with glycerol Viscosity, the more viscous a substance is.įigure 4.54: Pouring water versus pouring syrup. Viscosity Viscosity is a measure of how much a liquid resists flowing. The water flows much faster than the syrup or honey. Think how easy it is to pour water compared Viscosity is the resistance to flow of a liquid. 
The physical properties of organic molecules. In this chapter we will focus on the effects of van der Waals forces and hydrogen bonding on Lived force which holds the compounds together. Non-polar molecule leading to a weak, short These forces exist between dipoles and non-polar Molecules that are next to each other attract (remember that the electrons are always moving Usually evenly distributed but it is possibleĮlectrons might not be evenly distributed In non-polar molecules the electronic charge is One special case of this is hydrogen bonding:ĭipole-induced-dipole intermolecular forces Molecule, the positive pole of the one molecule will beĪttracted to the negative pole of the other, and the molecules When one dipole molecule comes into contact with another dipole An overview of theĭifferent types of intermolecular forces that are discussed in this chapter are Is slightly positive and the other is slightly negative. Is a molecule that has its charge unevenly distributed. 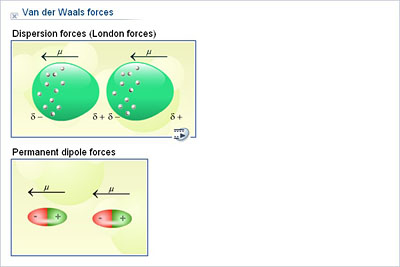
Intermolecular forces are forces that act between molecules. 
Holding molecules together we would not exist. The melting and boiling points of substances are. Intermolecular forces allow us toĭetermine which substances are likely to dissolve in other substances, and what The forces between molecules that bind them together are known as Iaszlo-photo, Fields of View and Capt' Gorgeous on Flickr. Note that we are showing two-dimensional figures when in reality theseįigure 4.52: Submicroscopic diagrams of the three phases of water. The more energy the particles have, the more likely they are to beĪble to overcome the forces that are holding them together. Remember also that the temperature of a material affects the energy The forces are very strong, the particles are held closely together in a solid Particles are able to move far apart since they are not held together very strongly. The forces, the more likely the substance is to exist as a gas. Substance is determined by how strong the forces are between its particles. You should remember from the kinetic theory of matter Properties, such as its phase, melting point andīoiling point. The types of intermolecular forces that occur in a substance will affect its physical Properties of organic molecules with their molecular structure. Have the learners research the safety data for various compounds, especially thoseīeing used in the experiments in this section, as a way of linking the 
4.4 Physical properties and structure (ESCKP) Physical properties and intermolecular forces (ESCKQ) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
